
Buoyant force
We've all seen it; you are part of an outdoor party or event and someone lets go of their balloon. Helplessly, all eyes glued on the balloon as it sails up, up and away.

Yes, this lone balloon represents a dear loss to someone. But even so, it raises is an interesting question – if gravity pulls everything down, why does the balloon go up?
It's a matter of buoyancy or buoyant force.
High pressure
Let's start with atoms. Everything we know is made of them. To quote the nerdy joke: "You can't trust atoms because they make up everything."
OK, seriously, although everything is made of atoms, not all atoms behave the same. In solid objects (bricks, tables, hair ribbons, baseballs, etc.) the atoms are held largely in place. However, in liquids and gases (water, air, lemonade, laughing gas, etc.) the atoms are much freer to wander about.
This is epic to today's tale.
Suppose one put a solid object (mirror, leaf, hot dog, etc.) in a liquid or gas (a vat of milk, seawater, carbon dioxide, etc.). Naturally, the atoms surrounding the solid would be bumping into it.
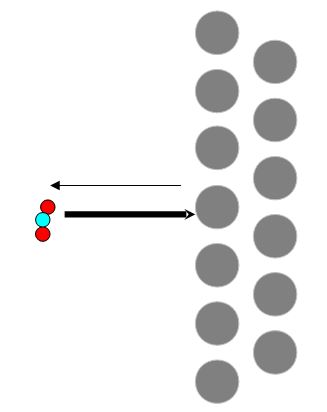
As they do. They'll bounce off the solid, the solid will be pushing back. Atoms, therefore, cause a pushing force on any object touching it. It is, however, most convenient to think of this force in terms of pressure.
No, we're not talking about sales pressure.

In science, pressure is the force per area. In everyday usage, pressure is expressed in "Pounds per square inch" (psi) – or how many pounds of force in on every square inch.
For example, you put air in your tires until the pressure gauge reads 35 psi. This means that the air in the tires pushes with the same force as a 35-pound child in every single square inch of space in the inside of the tire. Next time you get in your car look at the tires and think about this.
Amazing!
You raise me up
So, what can influence pressure? Many things actually; we shall focus on just one. Height. The force that a liquid or gas pushes on its surroundings must equal the weight it is being pushed down on because of gravity. So it shouldn't be a surprise that air pressure tends to decrease as you gain altitude (the air has less weight above it) any more than water pressure increases as you dive further below the surface (you get more water weight as you go down).
How is all this important?
Suppose you are swimming and your favorite can of whatever falls in. As you sadly see it sink below the water, the can will have pressure on it from the water acting on all its sides. But, and here's the punchline, the water pressure on the bottom side of the can will be more than the top.
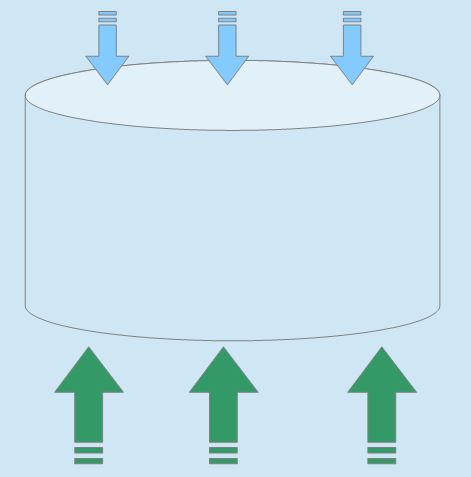
So the same process that causes pressure, combined with gravity, also produces a combined force directed upward. This is the buoyant force. The actual amount of force is described by Archimedes' Principle. In short, the buoyant force equals the weight of the liquid/gas replaced by the object itself. To break it down further:
The denser ("heavier" if you will) the liquid/gas the greater the force. We can see this in that air mattresses easily float on water but not in the air.
The volume of the object. In the case of the unfortunate can above, this can be explained because a larger can would either be wider (more area for the pressure to work) or taller (larger difference in pressure).
Bobbing along
Archimedes' principle has some peculiar consequences. Suppose I had a helium balloon.

Now suppose you also have a bowling ball of the same volume.

According to old Archimedes, both should have the same upward buoyant force. So how come the balloon will rise through the air but not the bowling ball?
The difference lies in their weight difference. True, the air will lift both with the same force, but it has a much harder job with the ball. The key to floating is then for an object to weigh less than the liquid/gas replaced by it.
Thus, the scientific explanation for flotation. Mind you, it remains for the serious investigator to explore this topic further through serious experimentation.

Fun for the family
A common demonstration of air pressure is the egg in a bottle demonstration (a video demonstration and explanation is here). The trick is to get a hard-boiled egg into a bottle even though the bottle opening is too small. In summary, the steps are as follows:
Wet the opening to the bottle as well as the part of the egg that will rest on the opening. This helps create a seal.
Get some paper, set it on fire, and place it in the bottle. Put the egg on top.
At first, the burning air will rise up and out of the top. The egg will wobble around a bit as this air pushes it around.
The fire will go out and the air in the bottle will go down in pressure.
The air outside now has a higher pressure than in the bottle. This difference will suck the egg into the bottle.
On the web
55-gallon steel drum can crush using atmospheric pressure
This video appears to be a class demonstration of what is a classic demonstration of air pressure – done on a large scale. The air in a 55-gallon steel drum is heated up and the hot air is allowed to escape. The hole in the drum is closed up and it is moved to a bath of cold water. After a little patience, the air in the can will cool and the pressure drops enough for the outside pressure to explosive crush the can. It is very amazing to see!
This video shows how a hot air balloon works.
Same deal as above, but now it is a submarine.
Don't miss out on future posts! Sign up for our email list and like us on Facebook!
Check out more hot topics, go back to Home Page
Comments? You can contact me at mailbox@thehomegrownprofessor.com